At Medistri, we understand that every laboratory result is only as reliable as the method behind it. Analytical method validation is the process by which a laboratory demonstrates, through documented and objective evidence, that a method is fit for its intended purpose. For medical device manufacturers, validated methods are not optional: they are a regulatory expectation across ISO/IEC 17025, Good Laboratory Practice (GLP), and Good Manufacturing Practice (GMP) frameworks. This article provides an overview of how Medistri approaches method validation, the key parameters we evaluate, and why this work matters for the safety and compliance of your products.
Why Method Validation Matters
Medical device testing generates data that directly informs regulatory decisions — from chemical characterization studies under ISO 10993-18 to cleaning validation and residual analysis. If the underlying analytical method has not been formally validated, the reliability of that data cannot be demonstrated to regulators, notified bodies, or auditors.
Method validation provides documented assurance that an analytical method consistently produces results that are accurate, precise, and reproducible under defined conditions. It also establishes the boundaries within which a method can be used — the concentration range, the matrix types, and the limits below which results should not be reported for quantitative purposes.
Regulatory and Standard Framework
Medistri’s validation approach is aligned with internationally recognized standards and guidance documents:
- ISO/IEC 17025: The international standard for testing and calibration laboratory competence. It requires that laboratories validate non-standard methods, laboratory-developed methods, and standard methods used outside their intended scope. Within a Type C accreditation scope, the laboratory demonstrates its competence to develop and validate analytical methods within a defined technology family.
- FDA Guidelines for the Validation of Chemical Methods (3rd Edition, 2019): This guidance defines concentration-dependent acceptance criteria for recovery, repeatability (RSDr), and reproducibility (RSDR), which Medistri applies as quantitative benchmarks throughout its validation studies.
- GLP (OECD Principles of Good Laboratory Practice): For studies supporting safety assessments, GLP requires that analytical methods used are validated and that all changes are controlled and documented. Medistri’s validated methods may be used to support GLP studies, provided that the validated conditions are respected and applied without modification or under controlled change management.
- GUM (Guide to the Expression of Uncertainty in Measurement): Provides the framework for estimating measurement uncertainty — a mandatory component of any validated quantitative method.
Key Validation Parameters
Each validation study at Medistri follows a structured experimental plan. The parameters evaluated represent the minimum validation requirements applicable to all methods developed under a given technology family. The following parameters form the core of every validation:
System Suitability Testing (SST)
Before any validation data is generated, the analytical system must demonstrate that it is operating within acceptable performance limits. SST verifies instrument and column performance as well as method-specific integration performance under routine conditions. Acceptance criteria are defined for each SST component and must be fulfilled before proceeding with the validation.
Linearity
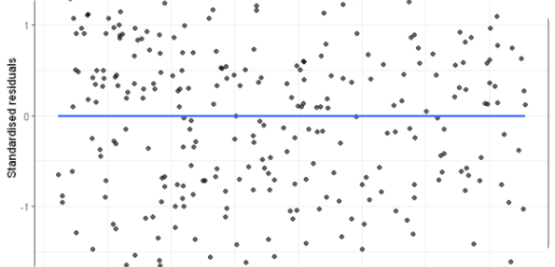
Linearity is evaluated by analyzing calibration standards covering the intended concentration range over multiple days, each prepared from freshly made working solutions. The response function is assessed through the coefficient of determination (R² ≥ 0.990), the coefficient of correlation (R ≥ 0.995), and visual inspection of the residuals plot to confirm the absence of systematic curvature.
Limit of Detection (LOD) and Limit of Quantification (LOQ)
The LOD represents the lowest amount of analyte that can be reliably detected, while the LOQ is the lowest concentration at which quantitative determination is achieved with suitable precision and accuracy. At Medistri, LOD and LOQ are determined using two complementary approaches: a signal-to-noise ratio method based on blank measurements (S/N ≥ 3 for LOD, ≥ 10 for LOQ), and a confirmation approach based on replicate measurements at the lowest standard concentration. Results below the established LOQ are not reported for quantitative purposes.
Instrumental Repeatability
Repeatability assesses the closeness of results obtained under the same operating conditions — same operator, same instrument, same day. It is evaluated by analyzing replicates at multiple concentration levels across multiple analytical series. The relative standard deviation (RSD) at each level must fall within the concentration-dependent acceptance limits defined by the FDA guidelines.
Intermediate Precision
Intermediate precision measures the variability of results when conditions are deliberately changed — different operators, different days, and different primary solution preparations. The experimental design involves a minimum of two operators each performing multiple series on different days, ensuring statistical relevance in accordance with ISO 5725 principles. The RSD values must remain within the reproducibility limits (RSDR) specified by the FDA guidelines for each concentration level.
Accuracy
Accuracy evaluates the degree of agreement between the measured result and the true value. It is determined by calculating the percentage recovery across all concentration levels used in the repeatability and intermediate precision studies. Recovery values must fall within the concentration-dependent acceptance ranges defined by the FDA guidelines — for example, 80–110% recovery at the 1–10 ppm level, with tighter ranges at higher concentrations.
Stability of Standards and Solutions
The stability of standard solutions under defined storage conditions is evaluated over a period reflecting the maximum anticipated storage time under routine laboratory conditions. Aliquots are analyzed at defined intervals, and results are assessed against the method’s established uncertainty to confirm that no significant degradation occurs within the validated storage period.
Measurement Uncertainty
Measurement uncertainty estimation is a mandatory requirement under ISO/IEC 17025. Medistri’s approach follows GUM principles: identifying sources of uncertainty (standard preparation, accuracy, repeatability), calculating standard uncertainties for each source, combining them, and applying a coverage factor (k=2) for a 95% confidence level to determine the expanded uncertainty. The expanded uncertainty must remain within predefined acceptance criteria — typically ≤ 25% for trace-level quantification.
How a Validation Study Is Conducted at Medistri
Every validation begins with a formal validation protocol that defines the scope, the analytical method, the experimental plan, and the acceptance criteria before any laboratory work is initiated. The protocol is reviewed and approved by the technical responsible, quality engineering, the quality manager, and the laboratory manager. The experimental work is then carried out following the defined plan: two operators perform analyses across multiple series on different days using freshly prepared solutions. All critical equipment is calibrated and qualified prior to use, and sample preparation and instrumental analysis are performed in designated, identified laboratory areas to minimize the risk of cross-contamination — in accordance with internal SOP requirements and GLP principles.
Results are compiled into a method validation report, which documents all experimental data, statistical evaluations, conclusions for each parameter, and any deviations or out-of-specification results. The report includes a summary table presenting the outcome of each validation test against its acceptance criteria, and concludes with a formal declaration of conformity.
Ensuring GLP Compliance
Medistri’s validation framework is designed to support both ISO/IEC 17025 accredited testing and GLP-regulated studies. Several elements of the validation process directly address GLP requirements:
- Identified and dedicated testing areas are used for different stages of the study — sample preparation in a designated preparation area, and instrumental analyses in a dedicated analytical area — ensuring proper environmental control.
- The Quality Assurance Unit operates independently from the study conduct and may audit the validation documentation to confirm its suitability for GLP study support.
- All changes to validated methods are classified as minor or major based on their potential impact on analytical performance. Minor changes (such as equivalent consumables or column replacement with identical specifications) are justified and documented. Major changes (such as a change of analytical principle or calibration strategy) require prior assessment and may trigger partial or full revalidation.
- For GLP studies specifically, all changes must be approved before implementation and documented in accordance with GLP requirements.
Ongoing Method Verification Through Proficiency Testing
Validation is not a one-time event. Once a method is validated and placed into routine use, its ongoing performance must be monitored. Medistri participates in proficiency testing schemes (PTS) and interlaboratory comparisons (ILC) to continuously verify the competence of its accredited test methods. Proficiency testing providers used by Medistri are accredited according to ISO/IEC 17043. Results are evaluated using z-scores calculated in accordance with ISO 13528: a z-score of |z| ≤ 2.00 is considered satisfactory, values between 2.00 and 3.00 are questionable, and |z| ≥ 3.00 is unsatisfactory. Each proficiency test result is verified internally and documented in a compliance report signed by the responsible technical personnel and quality department.
Why This Matters for Your Products
For medical device manufacturers, working with a laboratory that follows a rigorous, documented validation approach means that the analytical data supporting your regulatory submissions is defensible and traceable. Whether the data is used for chemical characterization under ISO 10993-18, cleaning validation, residual solvent testing, or extractables and leachables analysis, the underlying method must be validated to the standards expected by regulatory authorities and notified bodies.
Medistri’s validation framework — aligned with ISO/IEC 17025, FDA guidelines, GLP principles, and GUM — provides the scientific foundation that manufacturers need to support product safety and regulatory compliance throughout the device lifecycle.
Learn more about our laboratory services at our website here. To ensure your products meet the highest quality and safety standards, contact our dedicated team at contact@medistri.com.
— The Medistri Team
#Medistri
Medistri is Europe's leading independent contract sterilization company, founded in 2006 and headquartered in the heart of Switzerland. We are a global player providing essential infrastructure that powers healthcare innovation worldwide.
We serve customers of all sizes, from startups, university projects, and research institutes to Fortune 500 corporations, across Medical Devices, Pharmaceutical Packaging, and Biotech.
Medistri supports products at every stage of the lifecycle: pre-clinical development, go-to-market, and full industrialization.
Our fully integrated, end-to-end in-house solution allows our customers to focus on what matters most: engineering, innovation, and development, while we manage the critical back-end processes.
Our synchronized departments work together to provide a complete stack of services:
- Contract Sterilization & Validation Services
- Contract Laboratory Services
- Contract Manufacturing Services
- Logistics Services
- Consulting Services
We help you accelerate time-to-market, reduce barriers to scale, and deliver safer, more sustainable healthcare solutions to patients around the world.
Medistri’s Solutions for Products in Pre-Clinical Phase
During the pre-clinical phase, execution is essential to validate concepts, generate reliable data, and establish a strong foundation for regulatory approval.
Medistri serves as your integrated infrastructure partner, delivering a comprehensive portfolio of in-house Solutions optimised for R&D and proof-of-concept development.
Our solutions encompass R&D treatment cycles (custom sterilisation protocols), cleaning & reprocessing, product assembly, and packaging, alongside specialised testing including biocompatibility, extractables & leachables, sterile barrier integrity, environmental conditioning, laboratory analysis development, batch release, and regulatory consulting.
This vertically integrated model minimises vendor coordination, expedites development iterations, guarantees data integrity and compliance from inception, and enables your team to prioritise core scientific and engineering advancements.
Positioning you for seamless progression to clinical trials and commercialization.
Frequently Asked Questions